|
1/29/2024 0 Comments Covid vaccine side effect rash This data helped predict how well the vaccines would work for younger people. 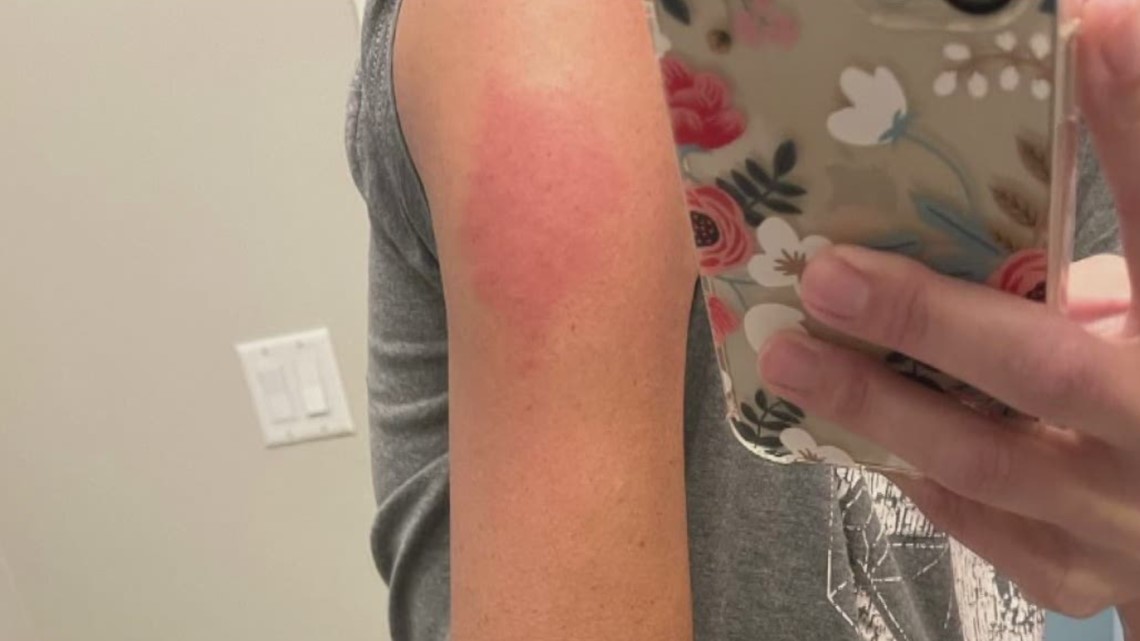
In December 2020, the Pfizer-BioNTech COVID-19 vaccine two-dose series was found to be both safe and 91% to 95% effective in preventing COVID-19 infection in people age 18 and older. That strain began spreading at the end of 2019. This vaccine was first tested against the original strain of the COVID-19 virus. 
Vaccines with FDA emergency use authorization or approval include:Ģ023-2024 Pfizer-BioNTech COVID-19 vaccine. In September and October 2023, the FDA authorized the use of the updated 2023-2024 COVID-19 vaccines made by Novavax, Moderna and Pfizer-BioNTech. The vaccines were changed to target a strain of the virus that causes COVID-19 called XBB.1.5. In June 2023, the FDA directed vaccine makers to update COVID-19 vaccines. Both included the original and omicron variants of the virus that causes COVID-19. In August 2022, the FDA authorized an update to the Moderna and the Pfizer-BioNTech COVID-19 vaccines. But the data still has to show that the vaccines are safe and effective. To provide vaccines sooner, the FDA gave emergency use authorization to COVID-19 vaccines based on less data than is typically required. Food and Drug Administration's (FDA's) vaccine approval process can take years.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
